AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Conjugate base3/6/2023  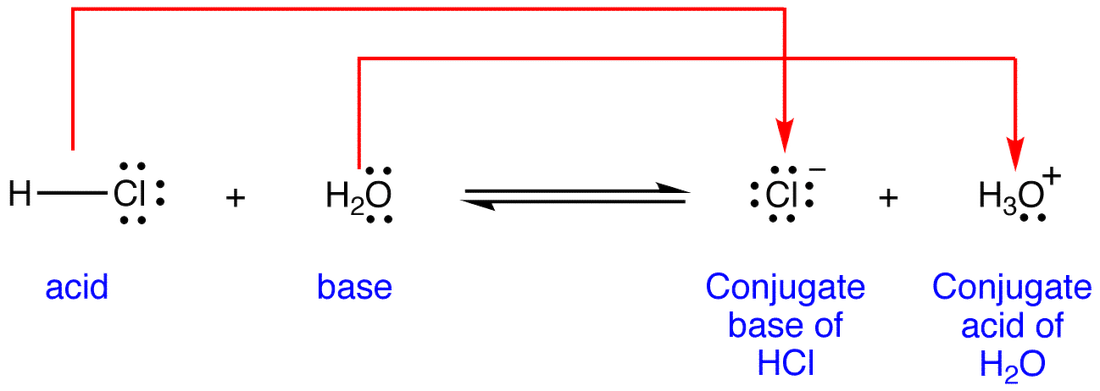
Therefore, acids and bases are electrolytes, with strong acids and bases behaving as strong electrolytes versus weak acids and bases behaving as weak electrolytes. Produce carbon dioxide when reacted with carbonatesĬommon examples: lemons, oranges, vinegar, urine, sulfuric acid, hydrochloric acidĬommon examples: soap, toothpaste, bleach, cleaning agents, limewater, ammonia water, sodium hydroxideĪcids and bases in aqueous solutions will conduct electricity because they contain dissolved ions. Produce hydrogen gas when reacted with metals AcidsĪre colourless when placed in phenolphthalein (an indicator)Īre pink when placed in phenolphthalein (an indicator)Īre red on blue litmus paper (a pH indicator)Īre blue on red litmus paper (a pH indicator) Table 5.1.1.General Properties of Acids and Bases. Acids and bases can be defined by their physical and chemical observations in the table below. They have opposing chemical properties and are able to neutralize one another to form H 2O, which will be discussed later in a subsection. The significance of hydrogen was reemphasized in 1884 when Svante Arrhenius defined an acid as a compound that dissolves in water to yield hydrogen cations (now recognized to be hydronium ions) and a base as a compound that dissolves in water to yield hydroxide anions.Īcids and bases are common solutions that exist everywhere. Around that same time, Joseph Louis Gay-Lussac concluded that acids are substances that can neutralize bases and that these two classes of substances can be defined only in terms of each other. In 1815, Humphry Davy contributed greatly to the development of the modern acid-base concept by demonstrating that hydrogen is the essential constituent of acids. In the eighteenth century, it was recognized that acids have a sour taste, react with limestone to liberate a gaseous substance (now known to be CO 2), and interact with alkalis to form neutral substances. When Robert Boyle characterized them in 1680, he noted that acids dissolve many substances, change the colour of certain natural dyes (for example, they change litmus from blue to red), and lose these characteristic properties after coming into contact with alkalis (bases).  
5.1 – Acid-Base Definitions & Conjugate Acid-Base PairsĪcids and bases have been known for a long time. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
